Blog
11.May.2026
Gut-Health Stacks That Work: Designing Smart Synbiotic Formulas (Probiotics + Prebiotics + Postbiotics)
Gut health is no longer a niche category. It has become one of the most important pillars in modern supplement innovation, influencing everything from daily wellness positioning to premium condition-specific concepts. But as the category grows, so does the noise. Too many products still treat gut health as a simple probiotic count on a label, with little attention paid to strain selection, fiber pairing, shelf-life stability, delivery format, or realistic claims.
That is where smart synbiotic design stands out.
A modern gut-health formula should not be built around a single hero ingredient. It should be built as a system. Probiotics, prebiotics, and postbiotics each play different roles, and the best formulas are designed to make those roles work together. The goal is not just to create a product that looks strong on paper. The goal is to create one that remains stable, manufacturable, consumer-friendly, and aligned with what can actually be supported on-pack and in market communication.
This is why synbiotic formulas are getting so much attention. When done well, they allow brands to move beyond basic gut supplements and toward more thoughtful, more targeted, and more credible product design. But doing them well requires more than combining trendy ingredients. It requires category understanding, strain logic, formulation discipline, and a clear view of where science ends and marketing should stop.
Which strains are being used, and why? Which fibers actually complement the probiotic species in the formula? Is the product meant for daily digestive comfort, microbiome support, or a broader wellness concept? Will the strains survive the intended shelf life in the chosen delivery format? Will the fiber system improve the formula, or create taste, texture, moisture, or process issues? And can the claims stay accurate without drifting into over-promise?
That is the real difference between a basic probiotic supplement and a well-designed synbiotic formula.
A synbiotic combines probiotics with substrates that support beneficial microbes, typically prebiotic fibers. A more advanced formula may also include postbiotics, which are non-living microbial preparations or metabolites that can contribute functional value without the same viability constraints as live organisms. When these three elements are matched correctly, the formula becomes more strategic, more robust, and often more commercially practical.
In other words, a smart gut-health stack is not just more complex. It is more intentional.
Different probiotic strains behave differently, tolerate processing differently, and fit different product concepts. Even within the same species, not all strains offer the same functionality, stability, or commercial value. That means a formula built only around a high CFU headline can miss what really matters: whether the strains chosen make sense for the intended product.
Strain selection should start with use case. A daily digestive formula, a travel-friendly product, a women’s wellness concept, and a child-focused gut supplement may all require different probiotic strategies. Some strains are selected for their historical commercial familiarity. Others are chosen because they are more resilient in formulation or better suited to a certain dosage format.
The second consideration is survivability. A strain may look attractive in supplier material, but if it struggles in the intended environment, the finished product may underdeliver by the end of shelf life. That is why manufacturers need to assess not only initial loading, but also real stability performance under practical storage conditions.
The third consideration is compatibility. Some strains work more smoothly with certain fibers, moisture environments, excipients, or capsule systems. Others are more sensitive and demand tighter formulation control.
This is why smart probiotic formulation is not a game of collecting as many strains as possible. A shorter, more rational formula often performs better than a crowded one.
CFU targets should be chosen based on the strain system, intended positioning, format constraints, and expected stability loss over time. A product does not become superior just because the front label shows a massive number. In some cases, inflated CFU targets are used to compensate for poor stability planning, rather than to improve real product performance.
The smarter question is not “How high can we go?” It is “What viable count do we want consumers to receive by the end of shelf life, and what does it take to get there responsibly?”
This matters because probiotics are living organisms, and live organisms do not behave like ordinary dry actives. Moisture, temperature, oxygen exposure, compression force, ingredient interactions, and packaging conditions can all influence viability. A manufacturer that understands this will build CFU targets backwards from finished-product reality, not forwards from label marketing.
That is especially important in global markets, where distribution conditions may vary significantly. A formula that looks stable in a controlled development environment may behave differently once exposed to real storage and logistics conditions.
So yes, CFU matters. But CFU without strain logic and stability planning is not a strong product strategy.
The right prebiotic can strengthen a synbiotic formula by helping support beneficial microbes, improving differentiation, and broadening the product story beyond live cultures alone. But fiber selection needs to be practical. Not every prebiotic works equally well in every formula, and not every one pairs well with every species or dosage form.
The key point is this: fiber selection should never be generic. It should match the probiotic system, the serving size, the format, the target audience, and the overall sensory strategy.
That does not mean postbiotics replace probiotics. It means they give brands another tool. In some formulas, postbiotics can complement live strains and help create a more advanced microbiome-support story. In others, they may be used to reduce dependence on ultra-sensitive live systems, especially in formats where stability is harder to maintain.
From a manufacturing perspective, postbiotics can be appealing because they are typically more robust during storage and processing. That makes them relevant in categories where live CFU preservation is difficult, or where a brand wants to create a more stable global product line.
From a positioning standpoint, postbiotics also help shift the gut-health conversation from “how many live organisms are inside” to “what functional system has actually been built.” That is a more mature direction for the category.
Still, postbiotics need just as much discipline in communication. They should not be used as buzzwords. Their place in the formula should be clear, deliberate, and aligned with the rest of the stack.
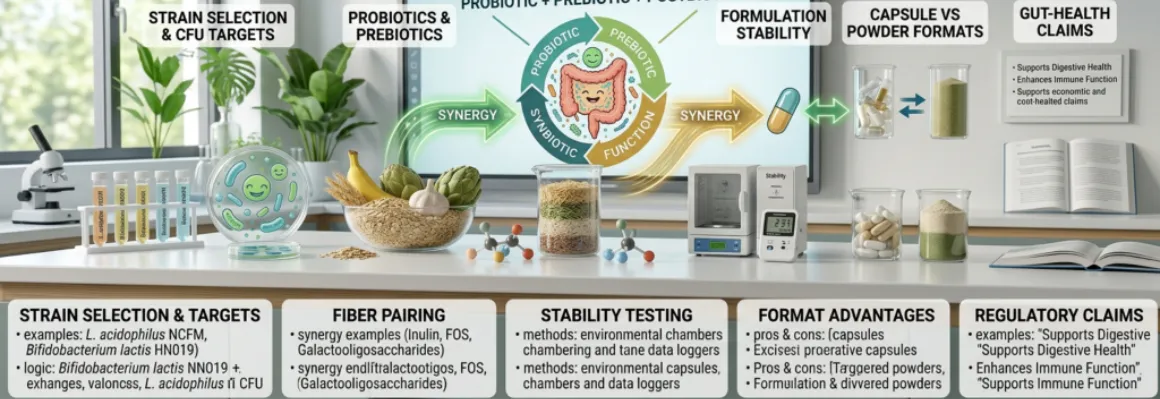
The first step is defining the purpose of the product. Is it a broad daily gut-health formula? A digestive comfort concept? A travel-friendly supplement? A beauty-from-within microbiome product? A children’s powder? A clinical-looking premium capsule? The right stack depends on the destination.
The second step is choosing probiotics with a realistic stability profile. Some strains are better suited to capsules. Others may be workable in sachets or sticks. Some require especially careful handling and packaging.
The third step is selecting fibers that support both the microbiome strategy and the product experience. A prebiotic may look ideal in theory, but if it makes the powder gritty, clumpy, or overly dense, that creates a commercial problem.
The fourth step is deciding whether postbiotics add genuine formulation value. In many cases, they do. But they should be included with purpose, not simply because the term is trending.
The result should be a formula where each component contributes to a coherent system rather than a label full of disconnected claims.
However, capsule space is limited. That becomes a challenge when combining meaningful levels of probiotics, prebiotics, and postbiotics into one product. If the prebiotic dose is intended to be substantial, a capsule may become impractical unless the product uses multiple units per serving.
But powders introduce more formulation pressure. Moisture sensitivity, flavor, flowability, clumping, probiotic viability, and user experience all become more challenging. A gut-health powder that tastes unpleasant or cakes in storage will struggle no matter how strong the formula looks on paper.
This is why format choice should come early in development, not late. The intended format shapes what the formula can realistically become.
A product can test beautifully at the bench stage and still fail commercially if the live counts drop too sharply over time. This is especially true for probiotic formulas exposed to temperature fluctuation, humidity, or packaging systems that do not provide adequate protection.
Stability planning needs to include more than the probiotic supplier’s starting specification. It should account for the full product matrix, manufacturing conditions, transportation realities, packaging barrier properties, and how the formula behaves over the intended shelf life.
Prebiotics can also influence stability indirectly. Some fibers attract moisture more easily. Some affect powder density, flow, or caking. Some interact differently within multi-ingredient systems. Even postbiotics, while generally more robust, still need to be evaluated as part of the whole formula.
This is why experienced manufacturers build gut-health products with stability in mind from day one. They do not leave it as a final check after the formula is already locked.
Consumers respond strongly to microbiome language, digestive comfort positioning, and broader wellness stories linked to the gut. That creates temptation to promise too much. But responsible brands know that gut formulas should be marketed with precision, not exaggeration.
The safest and smartest approach is to focus on what the formula is designed to support, not to overstate what it will guarantee. This is especially important in synbiotic formulas, where the science may support a structure-function style narrative but not broad disease-like claims or inflated before-and-after expectations.
Clear product positioning builds more trust than dramatic language. It also gives brands more room to scale internationally, where regulatory boundaries may differ across markets.
In the long run, a credible gut-health brand wins by sounding more confident because it is more disciplined, not because it is louder.
A strong partner should understand strain selection, CFU management, fiber compatibility, dosage form constraints, packaging logic, and stability planning. Just as important, they should know when a formula looks attractive in concept but becomes unrealistic in production or shelf-life performance.
They should also be able to guide brands through practical tradeoffs. For example, should a formula prioritize higher fiber loading or simpler capsule convenience? Should it rely on live probiotics only, or incorporate postbiotics for added robustness? Should the product be positioned as a broad daily wellness concept or a more targeted gut-support formula?
These decisions shape not just the formula, but the product’s long-term success in market.
Probiotics, prebiotics, and postbiotics each have value, but the best synbiotic formulas are built by understanding how they work together in real products. That means selecting strains with purpose, setting CFU targets realistically, matching the right fibers to the right species and formats, planning for stability across shelf life, and staying disciplined on claims.
That is what makes a gut-health stack actually work.
For brands, this creates a real opportunity. The market is ready for synbiotic formulas that feel more advanced, more credible, and more practical than standard probiotic products. But winning that opportunity requires formulation thinking that goes deeper than ingredient trends.
In gut health, the strongest products are not just assembled. They are engineered.
That is where smart synbiotic design stands out.
A modern gut-health formula should not be built around a single hero ingredient. It should be built as a system. Probiotics, prebiotics, and postbiotics each play different roles, and the best formulas are designed to make those roles work together. The goal is not just to create a product that looks strong on paper. The goal is to create one that remains stable, manufacturable, consumer-friendly, and aligned with what can actually be supported on-pack and in market communication.
This is why synbiotic formulas are getting so much attention. When done well, they allow brands to move beyond basic gut supplements and toward more thoughtful, more targeted, and more credible product design. But doing them well requires more than combining trendy ingredients. It requires category understanding, strain logic, formulation discipline, and a clear view of where science ends and marketing should stop.

What Makes a Synbiotic Formula Smarter Than a Standard Gut Health Supplement?
A standard probiotic product often focuses on one number: CFU. That number may matter, but it does not tell the whole story. A smarter gut-health formula asks more important questions.Which strains are being used, and why? Which fibers actually complement the probiotic species in the formula? Is the product meant for daily digestive comfort, microbiome support, or a broader wellness concept? Will the strains survive the intended shelf life in the chosen delivery format? Will the fiber system improve the formula, or create taste, texture, moisture, or process issues? And can the claims stay accurate without drifting into over-promise?
That is the real difference between a basic probiotic supplement and a well-designed synbiotic formula.
A synbiotic combines probiotics with substrates that support beneficial microbes, typically prebiotic fibers. A more advanced formula may also include postbiotics, which are non-living microbial preparations or metabolites that can contribute functional value without the same viability constraints as live organisms. When these three elements are matched correctly, the formula becomes more strategic, more robust, and often more commercially practical.
In other words, a smart gut-health stack is not just more complex. It is more intentional.
Probiotics: Why Strain Selection Matters More Than a Big CFU Number
One of the most common mistakes in gut-health product development is treating probiotics as interchangeable. They are not.Different probiotic strains behave differently, tolerate processing differently, and fit different product concepts. Even within the same species, not all strains offer the same functionality, stability, or commercial value. That means a formula built only around a high CFU headline can miss what really matters: whether the strains chosen make sense for the intended product.
Strain selection should start with use case. A daily digestive formula, a travel-friendly product, a women’s wellness concept, and a child-focused gut supplement may all require different probiotic strategies. Some strains are selected for their historical commercial familiarity. Others are chosen because they are more resilient in formulation or better suited to a certain dosage format.
The second consideration is survivability. A strain may look attractive in supplier material, but if it struggles in the intended environment, the finished product may underdeliver by the end of shelf life. That is why manufacturers need to assess not only initial loading, but also real stability performance under practical storage conditions.
The third consideration is compatibility. Some strains work more smoothly with certain fibers, moisture environments, excipients, or capsule systems. Others are more sensitive and demand tighter formulation control.
This is why smart probiotic formulation is not a game of collecting as many strains as possible. A shorter, more rational formula often performs better than a crowded one.
CFU Targets: Bigger Is Not Always Better
Consumers often assume that more CFUs automatically mean a better probiotic. From a marketing standpoint, that assumption is easy to exploit. From a formulation standpoint, it is too simplistic.CFU targets should be chosen based on the strain system, intended positioning, format constraints, and expected stability loss over time. A product does not become superior just because the front label shows a massive number. In some cases, inflated CFU targets are used to compensate for poor stability planning, rather than to improve real product performance.
The smarter question is not “How high can we go?” It is “What viable count do we want consumers to receive by the end of shelf life, and what does it take to get there responsibly?”
This matters because probiotics are living organisms, and live organisms do not behave like ordinary dry actives. Moisture, temperature, oxygen exposure, compression force, ingredient interactions, and packaging conditions can all influence viability. A manufacturer that understands this will build CFU targets backwards from finished-product reality, not forwards from label marketing.
That is especially important in global markets, where distribution conditions may vary significantly. A formula that looks stable in a controlled development environment may behave differently once exposed to real storage and logistics conditions.
So yes, CFU matters. But CFU without strain logic and stability planning is not a strong product strategy.
Prebiotics: Matching the Right Fiber to the Right Formula
Prebiotics are often added to gut formulas as an afterthought. They should not be.The right prebiotic can strengthen a synbiotic formula by helping support beneficial microbes, improving differentiation, and broadening the product story beyond live cultures alone. But fiber selection needs to be practical. Not every prebiotic works equally well in every formula, and not every one pairs well with every species or dosage form.
Inulin
Inulin is one of the most recognizable prebiotic fibers in the category. It brings strong consumer familiarity and can support a clear digestive wellness story. It is especially useful when a brand wants a well-known prebiotic on-pack. However, inulin can create formulation tradeoffs, particularly in higher-dose applications. It may affect texture, sweetness perception, or gastrointestinal tolerance depending on dose and user sensitivity.GOS
GOS is attractive because it is widely used in microbiome and digestive support concepts and fits well into more premium synbiotic positioning. It can be particularly useful when a brand wants a prebiotic with a more targeted or modern profile. That said, processing, hygroscopicity, cost structure, and label strategy all need to be considered.Apple Fiber
Apple fiber brings a different kind of value. It can support digestive-positioned concepts while also contributing to a more whole-food, natural ingredient story. In certain powder formats, it may offer branding advantages. But its sensory and textural effects need careful evaluation, especially in drinkable formulas where mouthfeel can make or break repeat purchase.Resistant Starch
Resistant starch is interesting because it supports a broader gut-health conversation while offering functionality that may fit certain powder systems well. It can be useful in formulas where the prebiotic story needs to feel more advanced or differentiated. However, like all fibers, it must be evaluated for process behavior, dosage practicality, and its effect on texture and dispersion.The key point is this: fiber selection should never be generic. It should match the probiotic system, the serving size, the format, the target audience, and the overall sensory strategy.
Postbiotics: The Stability-Friendly Layer of Modern Gut Formulas
Postbiotics are becoming a major point of interest because they allow formulators to build gut-health products with fewer viability constraints than live probiotics alone.That does not mean postbiotics replace probiotics. It means they give brands another tool. In some formulas, postbiotics can complement live strains and help create a more advanced microbiome-support story. In others, they may be used to reduce dependence on ultra-sensitive live systems, especially in formats where stability is harder to maintain.
From a manufacturing perspective, postbiotics can be appealing because they are typically more robust during storage and processing. That makes them relevant in categories where live CFU preservation is difficult, or where a brand wants to create a more stable global product line.
From a positioning standpoint, postbiotics also help shift the gut-health conversation from “how many live organisms are inside” to “what functional system has actually been built.” That is a more mature direction for the category.
Still, postbiotics need just as much discipline in communication. They should not be used as buzzwords. Their place in the formula should be clear, deliberate, and aligned with the rest of the stack.

Designing the Right Synbiotic Match
A good synbiotic formula is not built by adding one probiotic blend, one fiber, and one postbiotic ingredient into the same capsule and hoping the label looks advanced. The pieces need to fit together.The first step is defining the purpose of the product. Is it a broad daily gut-health formula? A digestive comfort concept? A travel-friendly supplement? A beauty-from-within microbiome product? A children’s powder? A clinical-looking premium capsule? The right stack depends on the destination.
The second step is choosing probiotics with a realistic stability profile. Some strains are better suited to capsules. Others may be workable in sachets or sticks. Some require especially careful handling and packaging.
The third step is selecting fibers that support both the microbiome strategy and the product experience. A prebiotic may look ideal in theory, but if it makes the powder gritty, clumpy, or overly dense, that creates a commercial problem.
The fourth step is deciding whether postbiotics add genuine formulation value. In many cases, they do. But they should be included with purpose, not simply because the term is trending.
The result should be a formula where each component contributes to a coherent system rather than a label full of disconnected claims.
Capsule vs Powder: Format Changes the Formula
Delivery format matters more in gut-health supplements than many brands initially expect.Capsules
Capsules are often preferred for convenience, portability, and cleaner dose management. They work well for many probiotic-centered concepts, especially when the target user wants a simple daily routine. Capsules can also help protect sensory-sensitive ingredients from direct consumer experience, which is useful when the formula includes components that might taste unpleasant in powder form.However, capsule space is limited. That becomes a challenge when combining meaningful levels of probiotics, prebiotics, and postbiotics into one product. If the prebiotic dose is intended to be substantial, a capsule may become impractical unless the product uses multiple units per serving.
Powders
Powders offer more room for fiber inclusion and can support more comprehensive synbiotic concepts. They are especially useful when the prebiotic is meant to play a significant role, rather than a token one. Powders also allow brands to create more lifestyle-friendly formats such as sachets, sticks, or daily drink mixes.But powders introduce more formulation pressure. Moisture sensitivity, flavor, flowability, clumping, probiotic viability, and user experience all become more challenging. A gut-health powder that tastes unpleasant or cakes in storage will struggle no matter how strong the formula looks on paper.
This is why format choice should come early in development, not late. The intended format shapes what the formula can realistically become.
Shelf-Life Stability: Where Good Gut Formulas Often Fail
Shelf life is one of the biggest make-or-break issues in synbiotic development.A product can test beautifully at the bench stage and still fail commercially if the live counts drop too sharply over time. This is especially true for probiotic formulas exposed to temperature fluctuation, humidity, or packaging systems that do not provide adequate protection.
Stability planning needs to include more than the probiotic supplier’s starting specification. It should account for the full product matrix, manufacturing conditions, transportation realities, packaging barrier properties, and how the formula behaves over the intended shelf life.
Prebiotics can also influence stability indirectly. Some fibers attract moisture more easily. Some affect powder density, flow, or caking. Some interact differently within multi-ingredient systems. Even postbiotics, while generally more robust, still need to be evaluated as part of the whole formula.
This is why experienced manufacturers build gut-health products with stability in mind from day one. They do not leave it as a final check after the formula is already locked.
Avoiding Over-Promising in Gut Health Claims
Gut health is one of the easiest categories to oversell, and one of the riskiest if brands are not careful.Consumers respond strongly to microbiome language, digestive comfort positioning, and broader wellness stories linked to the gut. That creates temptation to promise too much. But responsible brands know that gut formulas should be marketed with precision, not exaggeration.
The safest and smartest approach is to focus on what the formula is designed to support, not to overstate what it will guarantee. This is especially important in synbiotic formulas, where the science may support a structure-function style narrative but not broad disease-like claims or inflated before-and-after expectations.
Clear product positioning builds more trust than dramatic language. It also gives brands more room to scale internationally, where regulatory boundaries may differ across markets.
In the long run, a credible gut-health brand wins by sounding more confident because it is more disciplined, not because it is louder.
What Brands Should Look for in a Manufacturing Partner
Not every manufacturer is equipped to build advanced synbiotic products well.A strong partner should understand strain selection, CFU management, fiber compatibility, dosage form constraints, packaging logic, and stability planning. Just as important, they should know when a formula looks attractive in concept but becomes unrealistic in production or shelf-life performance.
They should also be able to guide brands through practical tradeoffs. For example, should a formula prioritize higher fiber loading or simpler capsule convenience? Should it rely on live probiotics only, or incorporate postbiotics for added robustness? Should the product be positioned as a broad daily wellness concept or a more targeted gut-support formula?
These decisions shape not just the formula, but the product’s long-term success in market.
Final Thoughts
The future of gut-health supplements belongs to smarter systems, not noisier labels.Probiotics, prebiotics, and postbiotics each have value, but the best synbiotic formulas are built by understanding how they work together in real products. That means selecting strains with purpose, setting CFU targets realistically, matching the right fibers to the right species and formats, planning for stability across shelf life, and staying disciplined on claims.
That is what makes a gut-health stack actually work.
For brands, this creates a real opportunity. The market is ready for synbiotic formulas that feel more advanced, more credible, and more practical than standard probiotic products. But winning that opportunity requires formulation thinking that goes deeper than ingredient trends.
In gut health, the strongest products are not just assembled. They are engineered.
Partner With Us
Contact us in the button below to further discuss about the possibility of using our best ingredients in your product line



















